About us
We provide researchers and corporates with support to secure the clinical efficacy
and safety of innovative medical devices.
 HOME
HOME About us
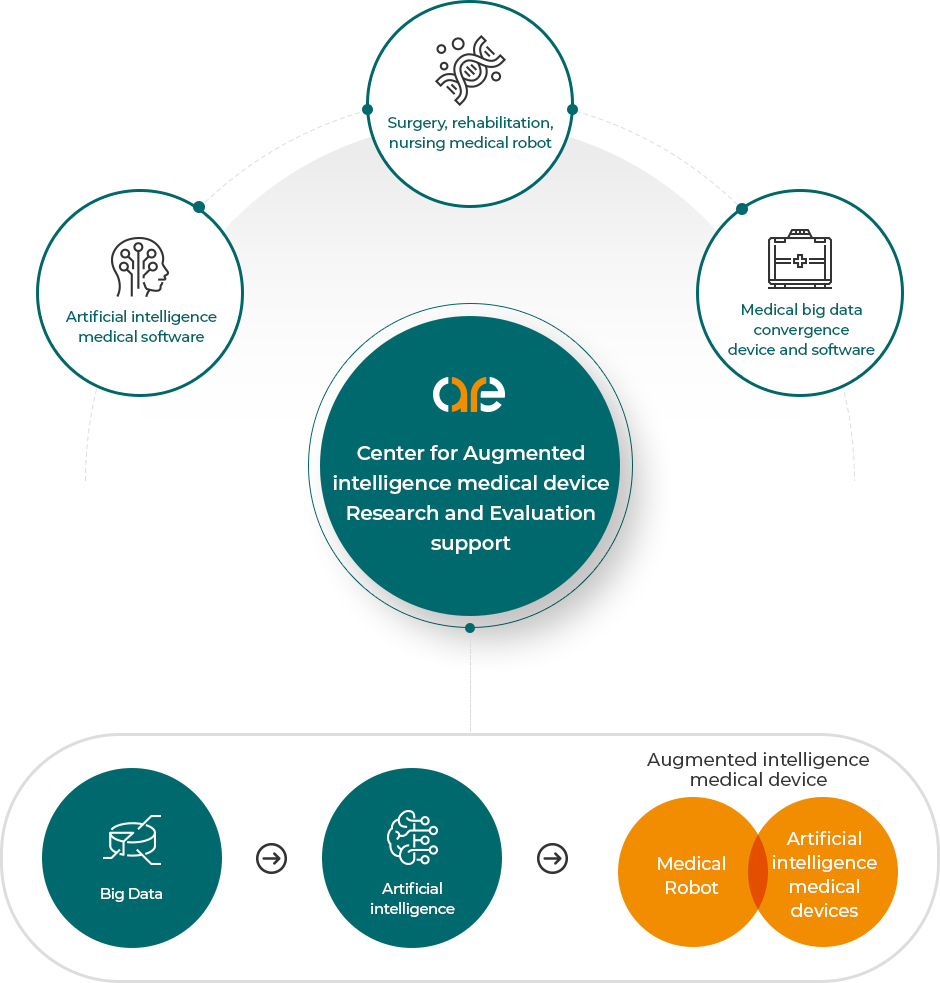
About us Medical Information Convergence Automated Device
Medical Information Convergence Automated Device
Medical Information Convergence Automated Device
1Convergence of medical information (clinical big data)
2Partial/full automation concept with active intelligence
It is necessary to implement a development method differentiated from existing medical devices and to secure clinical safety and efficacy
Onsite-based translational research with clinical experts and non-clinical/clinical trials for the development and commercialization of convergence advanced medical devices
It is necessary to construct an efficient infrastructure for open innovation.